2/1/2024
Setting the Trap
Dr. Sarah Jandricic

Thrips parvispinus has become a serious pest of tropical ornamental crops in North American since 2020. Due to the severity of the damage, Integrated Pest Management (IPM) strategies are needed to complement chemical control until an effective and economical biocontrol program can be developed for this pest.
Figure 1. Thrips parvispinus females swarming a yellow mandevilla flower. Most can be seen inside the flower tube feeding on nectar and pollen.
Mass trapping through traps, lures or trap plants can be an effective strategy for thrips. We’ve already seen that mass trapping cards really help with this pest (more on that later). And it seems that trap plants may also be an effective strategy when used properly in vegetative crops.
Background: Why try trap plants?
T. parvispinus, sometimes referred to as pepper thrips, has become a serious pest of tropical ornamentals such as mandevilla, Dipladenia, hoya, schefflera and anthurium since it was first discovered in Florida in 2020.
Although pesticides are available for this pest, active ingredients are limited in ornamentals. Not surprisingly, resistance has already been seen in this pest to Conserve (spinosad, Group 5), Delegate (spinetoram, Group 5) and Ference (cyantraniliprole, Group 28) in Ontario.
IPM Specialists in Ontario are making progress in developing effective biocontrol programs for T. parvispinus, but success with this pest is likely going to require a multi-pronged approach. Cultural controls, like trap plants, have proven effective in the past for western flower thrips and other pests. As T. parvispinus seems very attracted to flowers (Figure 1), this seemed like a good approach to try.

Tests of trap plants: What we did
Test were conducted in a commercial greenhouse in Ontario where T. parvispinus had previously been controlled with pesticides in the 2022 growing season, but where the growers were interested in more sustainable control methods in 2023.
Figure 2. A trap plant consisting of Garvinea Gerbera and sweet alyssum (left) in a crop of vegetative mandevilla (right).
In Summer 2023, we initially investigated the use of two flowering ornamentals as banker plants for Orius insidious, a known natural enemy of T. parvispinus. The idea here was to decrease the costs of using high levels of Orius by providing them with a pollen source, and encouraging establishment and reproduction in the crop. We chose Garvinea Gerbera (a long-lasting landscape plant) and Sweet Alyssum Gigi (a fast-growing plant) to see which would be more effective.
To do this, we filled 14-in. pots with seven sweet alyssum plugs each at the base of the pot and placed a large-flowering Garvinea Gerbera in the center of the same pot (Figure 2). We had 17 trap pots for a total of 950 crop plants (one trap plant for every 50 crop plants), spaced evenly down the length of the crop. However, it quickly became apparent that these plants were far better at acting as a trap plant for T. parvispinus than they were as banker plants for Orius, so we pivoted our objectives.
To evaluate these crops as trap plants, each week we tapped both the trap plants separately (as they occupied different space in the pots), as well as a subset of the mandevilla. This allowed us to compare T. parvispinus numbers on the different plants over time.
Were trap plants effective?
The short answer is yes!
The gerberas were not much more attractive than the crop itself until Week 4, so these didn’t make a very good trap plant (which is why we stopped monitoring them in Week 5). However, after two weeks of being placed in the crop (Figure 3), the sweet alyssum were already approximately two times more attractive to T. parvispinus than vegetative mandevilla. This is likely due to the large number of small flowers and high levels of pollen.
 Pictured: OMAFRA summer student Avery Johnson doing plant taps on trap plants in a commercial greenhouse.
Pictured: OMAFRA summer student Avery Johnson doing plant taps on trap plants in a commercial greenhouse.
By Week 3, thrips numbers on the alyssum had exploded. There were over four times more thrips on the trap plant than the mandevilla. We realized that at this point, the T. parvispinus could have completed a generation on the alyssum and could be acting as a source plant. This would explain why T. parvispinus numbers were also highest in the crops in Weeks 3 to 5. Sweet alyssum (Lobularia maritima) has not previously been reported as a host plant for T. parvispinus (i.e., a plant it can complete development on).
We confirmed that T. parvispinus were indeed reproducing on sweet alyssum with plant washes. These revealed a high proportion of larvae on the alyssum that weren’t coming out in our weekly plant taps.
To combat the trap plants becoming a potential source of thrips, rather than a sink, we did a spray of spinosad (Conserve) at label rates on just the trap plants. This has shown to be an effective way to use trap plants for western flower thrips .
Here, T. parvispinus numbers on the trap plants went down by 40% post-spray. Although this wasn’t the control we were hoping for, this was the first time the thrips numbers in the crop were lower than in the start of the experiment (Figure 3). This suggests more thrips might be moving from the crop onto these plants and being killed by the pesticide than evidenced by our plant taps.
On Week 6, we also compared mandevilla in blocks that had the trap plants to nearby blocks that didn’t (Figure 4). Although T. parvispinus numbers were lower everywhere on Week 6, this data confirmed that trap plants were having a significant effect. Mandevilla in blocks with the trap plants had an average of almost three fewer thrips per plant. Given that relatively low numbers of T. parvispinus cause economic damage compared to other thrips, even reductions of this magnitude are extremely important.
Since thrips numbers were still high on the trap plants after pesticide application (Figure 3), we bagged up the trap plants and replaced them with clean, vegetative alyssum. Interestingly, vegetative alyssum still proved to be three times more attractive than the crop plants when new trap plants were sampled Week 7. This means alyssum still shows promise as a trap plant in the winter when it tends not to flower.
Fi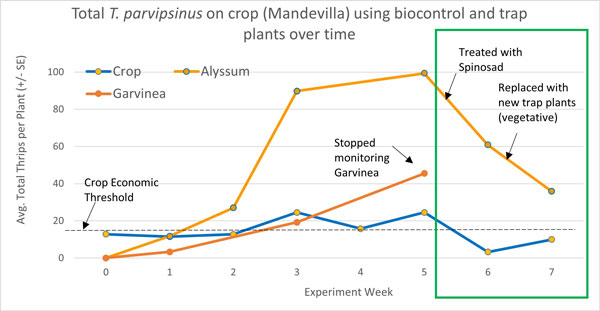 gure 3. Results of a seven-week on-farm trial comparing Thrips parvispinus populations on crop (mandevilla) versus trap plants (Garvinea and sweet alyssum). T. parvispinus were highly attracted to the sweet alyssum over the Garvinea and the mandevilla. Thrips numbers increased on the crop plants in Week 3, indicating trap plants might be acting as a source of thrips after two weeks. Once trap plants were sprayed and then replaced, thrips numbers on the crop reached their lowest levels in the experiment (green box).
gure 3. Results of a seven-week on-farm trial comparing Thrips parvispinus populations on crop (mandevilla) versus trap plants (Garvinea and sweet alyssum). T. parvispinus were highly attracted to the sweet alyssum over the Garvinea and the mandevilla. Thrips numbers increased on the crop plants in Week 3, indicating trap plants might be acting as a source of thrips after two weeks. Once trap plants were sprayed and then replaced, thrips numbers on the crop reached their lowest levels in the experiment (green box).
Caveats & recommendations
We’re currently testing this again over the winter to confirm that alyssum can be an effective trap plant for T. parvispinus in mandevilla year-long in the Northeastern area of North America. Although our trial has just started, it’s proving promising already (Figure 5).
You can see that after the trap plants were introduced numbers in the crop (9-in. mandevilla) started to slowly decline. Recent assessment of the trap plants on Week 44 showed seven times as many thrips on the alyssum versus the crop. We know from experience that the actual numbers of thrips hiding in the alyssum is much higher. The next step is to replace these with new, thrips-free trap plants and continue monitoring throughout the winter.
Based on this and the previous trial, here are some preliminary recommendations on the use of sweet alyssum trap plants for T. parvispinus:
■ A HIGH volume of trap plants is needed. In the first trial, we had a ratio of one trap plant for every 50 crop plants (both planted to fill 14-in. pots). The ratio is even higher in the new 9-in. pot trials. Before you endeavor down this pathway, ask yourself if you have the capacity to propagate large amounts of sweet alyssum in a CLEAN area free from T. parvispinus on a regular schedule.
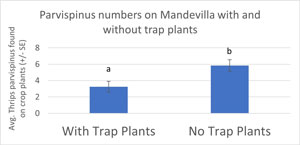 Figure 4. Average T. parvispinus numbers on crop plants (mandevilla) with and without trap plants. Thrips numbers were higher in the blocks of crop plants that didn’t use trap plants (control).
Figure 4. Average T. parvispinus numbers on crop plants (mandevilla) with and without trap plants. Thrips numbers were higher in the blocks of crop plants that didn’t use trap plants (control).
■ Trap plants MUST be thrown out and replaced every 14 to 17 days. Otherwise, you run the risk of these plants becoming a source of T. parvispinus, instead of a sink. Unfortunately, we’ve found spraying sweet alyssum doesn’t give great control, as coverage of this very “fluffy” plant is difficult. Drenches might be a more effective option, but this would require careful monitoring of efficacy. (Remember, systemic pesticides don’t translocate to petal tissue!)
■ Be mindful of how you’re disposing of your trap pants. It’s essential you bag them up as you go to prevent thrips from being disturbed and just flying back into the crop.
■ Note this concept is only showing promise in VEGETATIVE mandevilla so far. At this farm, any flowers on the crop are regularly being removed by workers to ensure the trap plant stays more attractive. A separate trial in flowering hibiscus (where flowering is constant, therefore removal is impossible) didn’t show sweet alyssum to be effective. The better option there was to put a large amount of sticky cards up near the hibiscus flowers.
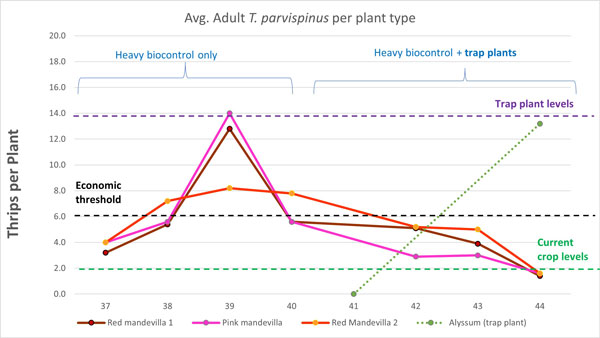 Figure 5. Results of a trial testing sweet alyssum alone as a trap plant in 9-in. vegetative mandevilla, starting in Week 37. The presence of trap plants is correlated with a slow reduction in T. parvispinus
Figure 5. Results of a trial testing sweet alyssum alone as a trap plant in 9-in. vegetative mandevilla, starting in Week 37. The presence of trap plants is correlated with a slow reduction in T. parvispinus
on the crop.
■ We’ve only tested one variety of sweet alyssum (Gigi). Other varieties might prove more attractive. This is something we hope to test later.
Take-home message
Sweet alyssum has the potential to be a powerful cultural control to reduce the number of T. parvispinus on tropical ornamentals. However, testing is in its initial stages. Trap plants are showing quite a bit of promise in mandevilla crops when vegetative or when flowers are regularly removed. High levels of trap plants are needed (one 14-in. pot full of sweet alyssum for every 50 pots of 9- to 12-in. mandevilla), and plants need to be thrown out and replaced every 14 to 17 days. It’s currently unclear if this tool can be used in flowering crops and if it’s economically feasible for all growers (requiring growing space and regular labor). GT
Dr. Sarah Jandricic is a Greenhouse Floriculture IPM Specialist for the Ontario Ministry of Agriculture, Food & Rural Affairs (OMAFRA).