3/1/2022
Technology in a Working Nursery
Stanton Gill
 In Part 1 of the drone series in the February issue of GrowerTalks
In Part 1 of the drone series in the February issue of GrowerTalks, we demonstrated to growers how drones are being used in greenhouse crops and in outdoor growing areas, such as container-grown chrysanthemums.
In Part 2, we look at using drones to apply low-risk materials in a working nursery setting to control bagworms (Thyridopteryx ephemeraeformis Haworth [Insecta: Lepidoptera: Psychidae]). In this trial, we used a spray drone at a wholesale nursery in central Maryland on field production of evergreen Thuja Green Giants hybrid trees that were 2-meters (6.5-ft.) tall.
Pictured: Drone operator Kirk Floyd getting the pesticide drone-sprayer ready. The drone used was a Model DJI Agras MG-1P.
Situation
Thyridopteryx ephemeraeformis (also called the evergreen bagworm, eastern bagworm, common bagworm, common basket worm or North American bagworm) can feed on over 50 families of deciduous and evergreen trees and shrubs. Severe infestations can damage the aesthetics and health of host plants, especially juniper (Juniperus) and arborvitae (Thuja). Thuja Green Giant is extremely popular among landscape installers and the nursery industry has responded by planting large blocks of Thuja Green Giant to meet this market demand.
Economic impact to the nursery and landscape industry
The wide host range, high female fecundity and method of dispersal by wind make bagworm a major problem in nurseries and in landscape settings. Damage to foliage can be extensive. Less than 10% of damage on woody plants is tolerated by consumers. During the summer months, as few as four bagworm larvae can cause a 4-ft. arborvitae to be unmarketable for sale.
Adult bagworms
Adult bagworms will often go unnoticed in the nursery and landscape, especially the female, because she’s enclosed in her bag and inside of her pupal casing throughout her life. The adult female’s wings and appendages are greatly reduced to vestigial mouthparts and legs, small eyes, and no antennae or wings. The female remains in a caterpillar-like state, mates and then the bag becomes essentially an egg-filled sac.
The male bagworm emerges as a free-flying moth that’s hairy and charcoal black. Neither the male nor the female adult feeds. The female lives a couple of weeks, while the male lives only one to two days. After mating, the female lays a large egg clutch (500 to 1,000 eggs) inside of her pupal case enclosed within her bag. The eggs are smooth and cylindrical in shape, and laid in a mass that’s covered in a waxy, tuft-like layer. Bagworm eggs will overwinter.
Bagworm larvae
Hatching larvae are small (under 2-mm long) and often disperse to surrounding plants by spinning a silken thread and “ballooning” on the wind. Once a suitable host is found, the caterpillar begins feeding and incorporating material into its bag, which it constructs with pieces of twigs, leaves and silk. Only the head and the thorax emerge from the anterior end of the bag so the caterpillar can feed and move along plant material.
Trial using a drone that sprays: Materials & methods
We applied reduced-risk products or biopesticides (Table 1) with a sprayer mounted on a drone (Model DJI Agras MG-1P). These insecticides were applied in two to three passes over rows of Thuja Green Giant. The rows were 120-ft. long and 4-ft. wide. Ten plants with a two-plant buffer between were randomly selected in each row for pre- and post-counts.
The trees were 2-meters (approximately 6-ft.) tall, a meter and a third (approximately 4-ft.) wide and planted on 2-meter (approximately 6-ft.) centers. Treatments were applied on July 7, 2021, and insecticide efficacy was evaluated as percent mortality on July 20 (13 days after treatment [DAT]).
 Pictured: Matthew Price (left), and Mollie Wyatt and Suzanne Klick (right) add bagworms to mesh bags placed over treated branches.
Pictured: Matthew Price (left), and Mollie Wyatt and Suzanne Klick (right) add bagworms to mesh bags placed over treated branches.
Bagworm neonates hatched from eggs collected in bags from last year’s untreated trees. These neonates fed on untreated arborvitae foliage in plastic observation cages until they reached 2nd to 3rd instar. Treatments were applied using the drone with the attached sprayer and allowed to dry for an hour. Then student interns added the bagworms to a mesh bag placed over the treated branches, confining the bagworm larvae to a treated branch. One end of the mesh bag was cable-tied closed before bagworms and a treated branch were encased inside the bag. The other end of the bag was cable-tied around the treated branch. The confined bagworms fed on the treated branches for 13 days before efficacy data were collected.
Branches with netted cable-tied bags were cut off treated trees and taken back to our research lab for post counts. Netted bags were opened, cutting off the cable-ties, and bagworms were placed in the middle of a circle drawn on a paper plate. Bagworms were given 10 minutes to crawl beyond the line of the circle. Those bagworms crawling outside the circle were considered alive and those that did not were considered dead.
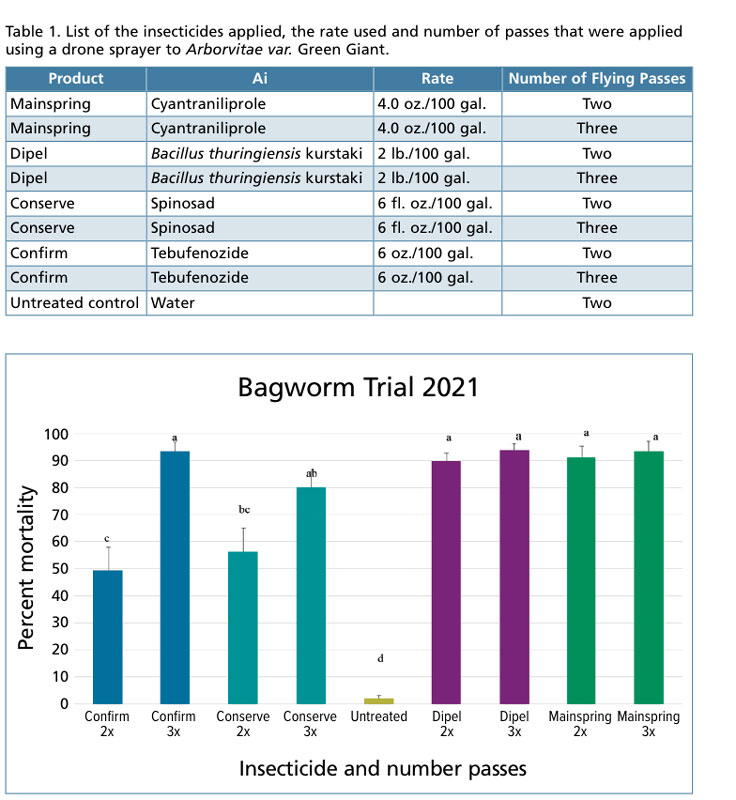
Discussion
• Having the drone go down the row and back again for double coverage gave fairly good control for treatments of Conserve and Confirm. Slightly better control was obtained with this same application method for Dipel and Mainspring.
• When we increased the number of passes the drone made on a treatment row to three passes with the spray drone, the efficacy of Confirm and Conserve significantly increased, likely due to better coverage. We didn’t see a significant increase in efficacy with Dipel and Mainspring when comparing two spray passes to three spray passes with the drone.
• All of the treatment gave significantly better control than the water-treated plants.
Where are we headed from here?
In 2022, we’ll continue to expand the number of low-risk pesticides we trial in nurseries. In Part 1, we used a fairly large drone to apply low-risk materials to chrysanthemums grown outdoors. In 2022, we’ll look at using smaller drones, more suited for use inside a working greenhouse. We’ll report on these results in 2023. GT
Special thanks for assisting in this field trial goes to: Brian Kunkel, Extension Specialist in IPM and Entomology, University of Delaware Extension; Suzanne Klick, LEAD Technician, University of Maryland Extension; Kirk Floyd, Drone pilot, KDrone Services; Matthew Price, Student Intern, University of Maryland Extension; and Mollie Wyatt, Student Intern, University of Delaware.
Stanton Gill is an Extension Specialist in IPM and Entomology at the University of Maryland Extension, and Central Maryland Research & Ed Center. He can be reached at Sgill@umd.edu.