7/1/2019
Whiteflies: A Major Problem That Just Doesn’t Go Away
Stanton Gill, Brian Kunkel, Suzanne Klick, Rachel Ross & Clarissa Hardcastle

In a prize fight, the last person standing is usually the winner. Whiteflies are like a boxer who keeps getting knocked down with brutal head and torso punches, but comes back up for one more round and wins in the end. We may hurt it with our pesticides, but not inflict a knockout—kind of a “Rocky” movie situation.
Historically, whiteflies were one of the first insect problems encountered by English growers in hothouses back in the late 19th century. They imported the pest from South and Central America, and it thrived in the new hothouses the wealthy English were constructing. More than a century later, we’re still trying to conquer this pest group. Whiteflies have been hit with just about every pesticide over the last 100-plus years.
Since this straight pesticide control strategy has major flaws, many growers are investigating an integrated approach of using pesticides that potentially work better with biological control. Several of the major chemical companies are developing new classes of pesticides that have greater potential for use in this integrated approach and most of these are systemic materials. In 2018, we worked with several of the major companies, including Syngenta, BASF, Bayer and Vestaron.
Our trials
Clean-rooted poinsettia plants were obtained from Ball Horticultural Company and transplanted into 6-in. pots. Ten completely randomized blocks of poinsettias were used in our trial. The plants were maintained as if in a production greenhouse with regards to lighting and fertilization throughout our experiment. Plants infested with Bemisia tabaci (B-biotype) were brought into the greenhouse to infest the poinsettias used in our insecticide efficacy trials.
In the early stages of the trial, the whitefly pressure was light, but as the fall progressed, the whitefly populations began to exert feeding and egg-laying pressure on all of the plants. Insecticide efficacy, or lack of efficacy, became evident as the pest pressure increased through the final stages of the trial.
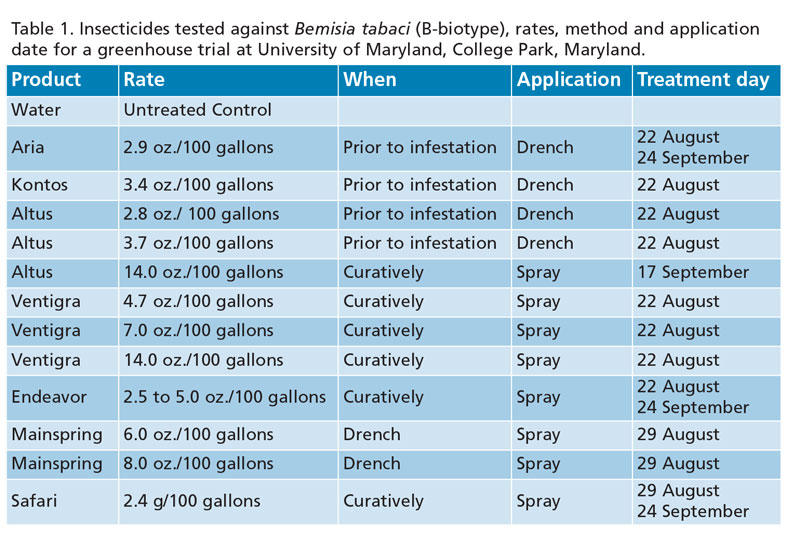
Our experiment evaluated the insecticides Aria, Kontos, Altus, Ventigra, Endeavor, Mainspring and Safari against water as the control. (Treatment dates, method of treatment, frequency and rate of insecticide are provided in Table 1.)
Drench applications were made prior to poinsettia exposure to whiteflies. Efficacy was determined by counting the number of live nymphs or adults found on a lower and upper leaf from each treated plant. Adults were counted immediately after leaf removal and nymphs were counted with a dissecting scope.
Results
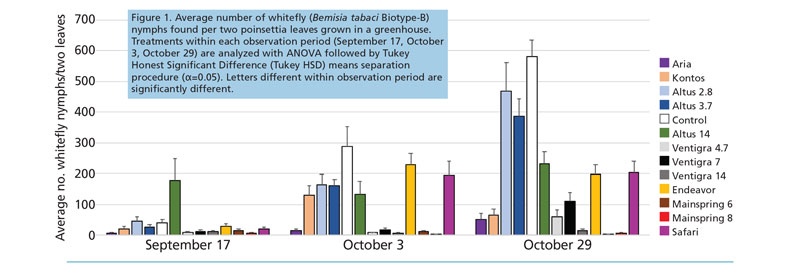
Nymphs (Figure 1). The first evaluation (September 17) found leaves infested with up to about 50 eggs per two leaves on each treated plant. Mainspring at the 8-oz. rate was the only insecticide with significantly fewer nymphs than the untreated control on September 17
(F=4.1; df=12,96; P<0.0001). Aria, Kontos, Altus 14-oz. spray; all Ventigra treatments; and both Mainspring treatments had significantly fewer nymphs present on the October 3 evaluation. However, the Kontos and Altus 14-oz. treatment still had over 100 nymphs per two leaves, whereas the other treatments with significantly fewer than control plants had less than 20 nymphs (F=11.0; df=12,108; P<0.0001).
The final evaluation (October 29) found the Safari, Endeavor, Altus 3.7-oz. and 14-oz. treatments with significantly fewer nymphs than the control; however, both treatments had over 200 nymphs per two leaves. Aria, Kontos, Ventigra treatments and Mainspring treatments were significantly different than untreated controls (F=22.6; df=12,106; P<0.0001) and had nymph populations less than 100 per two leaves. Mainspring treatments had numerically fewer nymphs found on two leaves than other treatments.
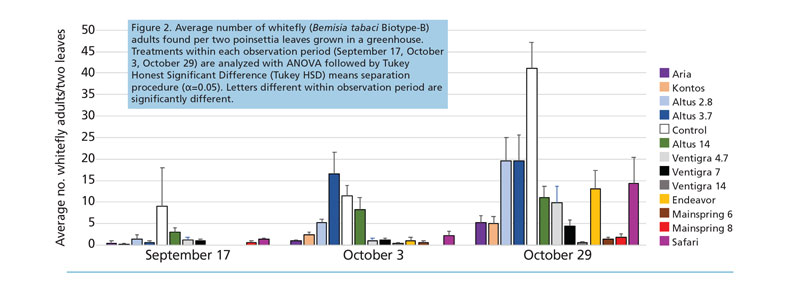
Adults (Figure 2). There were no significant differences between the treatments with how many adults were found on plants during the September 17 evaluation (P>0.05). All of the treatments had significantly fewer adults compared to the untreated control except the Altus treatments (F=7.9; df=12,108; P<0.0001).
The final evaluation found all treatments with significantly fewer adults compared to the untreated control (F=8.5; df=12, 106; P<0.0001). Ventigra 14-oz. treatment and Mainspring treatments had numerically fewer adults than any other treatment.
Summary
Mainspring and Ventigra consistently performed better than other products tested in our efficacy trial against whiteflies infesting poinsettias. Altus applied at the 14-oz. rate as a foliar application significantly reduced the number of live nymphs in our trial; however, the population found was still about 100 nymphs or more per two leaves.
Aria and Kontos significantly lowered the number of nymphs compared to untreated control and populations were fewer than 100 nymphs per two leaves. We observed a number of parasitoids, including adult Eretmocerus eremicus—parasitoid of whitefly sessile stages—flying among or walking on the poinsettias, thus, we feel these products have potential for a combination of chemical and biological control for dealing with whiteflies.
Stanton Gill is Extension Specialist in IPM for Greenhouses and Nursery for the Central Maryland Research & Education Center, and University of Maryland Extension and Professor at Montgomery College. He can be reached at Sgill@umd.edu or (410) 868-9400. Brian Kunkel is an Extension Ornamental IPM Specialist for the University of Delaware Cooperative Extension. He can be reached at bakunkel@udel.edu or (302) 893-6077. Suzanne Klick is Lead Technician at the Central Maryland Research and Education Center for the University of Maryland. Rachel Ross is a Student Intern at the University of Maryland. Clarissa Hardcastle is a Student Intern at the University of Delaware.