5/1/2019
Ways to Manage Mealybugs
Stanton Gill, Brian Kunkel, Suzanne Klick & Rachel Ross
The worst nightmare of greenhouse growers or a conservatory manager is mealybugs in the greenhouse. Once this pest is established, growers are guaranteed to spend endless hours and lots of money trying to get rid of this pest group.
Since this pest doesn’t fly (at least the females don’t fly), then it’s usually introduced on infested plant material. Since so many growers bring in cuttings from tropical sites, you have to be very watchful not to introduce this horrendous pest into your house.
Citrus mealybugs have been collected from at least 27 host plant families. Many ornamental plants grown in greenhouses are susceptible to attack, including begonia, coleus, amaryllis, cyclamen, poinsettia and dahlia. Plants such as hibiscus, Boston ferns and mandevilla shipped in from the south are usually excellent sources of citrus mealybug populations.
Mealybugs damage hosts by sucking out plant sap, by excreting honeydew in which sooty mold can grow, and by causing distorted growth and premature leaf drop with their toxic saliva. They further disfigure plants by secreting cottony wax. Infested plants usually die unless the pest is controlled.
The difficulty of controlling citrus mealybugs
Several natural enemies have been used effectively at controlling low to medium populations of citrus mealybug. Leptomastidea abnormis (Girault), Leptomastix dactylopii Howard, Chrysoplatycerus splendens Howard and Anagyrus pseudococci (Girault) are common wasps parasitic of second and third instar nymphs. Common predators include brown lacewing, Sympherobius barberi (Banks) and green lacewing, and Chrysopa lateralis Guérin.
Chemical control of citrus mealybugs is often an inefficient management strategy due to their habit of hiding in crevices between foliage and fruit. A powerful force pump and penetrating contact insecticide can be used to control mature insect populations. Applications of contact materials made before spring flush have been found to be the most effective strategy for citrus mealybug management.
In 2018, working with Syngenta, BASF and Bayer, we evaluated several new classes of insecticides for citrus mealybug. Most of these new materials are systemic insecticides that are more likely to carry into parts of the plant that mealybug feed.
We conducted two trials: In the first, we started with clean coleus plants and brought in an infestation of citrus mealybug. This trial evaluated the efficacy to keep mealybug populations from establishing.
In the second trial, we used previously infested plants in which pre-counts were taken before treatments. Treatments were applied to established populations on coleus and evaluated after 22 and 58 days after treatment (DAT).
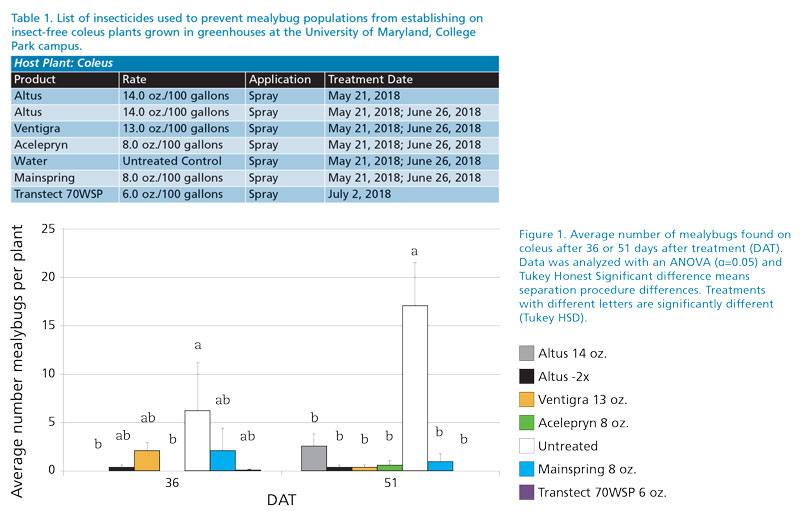
First trial
Treatments (Table 1) were made at College Park, Maryland, at university managed greenhouses on May 21, 2018. Evaluation of product efficacy occurred on June 26 (36 DAT) and July 11 (51 DAT) with 10 replicates.
The number of mealybugs found per plant was used to indicate how well an insecticide prevented mealybug population from establishing. Mealybugs were able to establish significant populations on untreated plants compared to some treatments (Figure 1). Signifi-cantly fewer mealybugs were found on plants receiving a single application of Altus or Ventigra at 36 DAT (F-value= 3.1; df=6,54;P=0.0116). However, by 51 DAT, all treated plants had significantly fewer mealybugs on them compared to the control (Figure 1; F-value= 21.4;df=6,54;
P<0.0001).
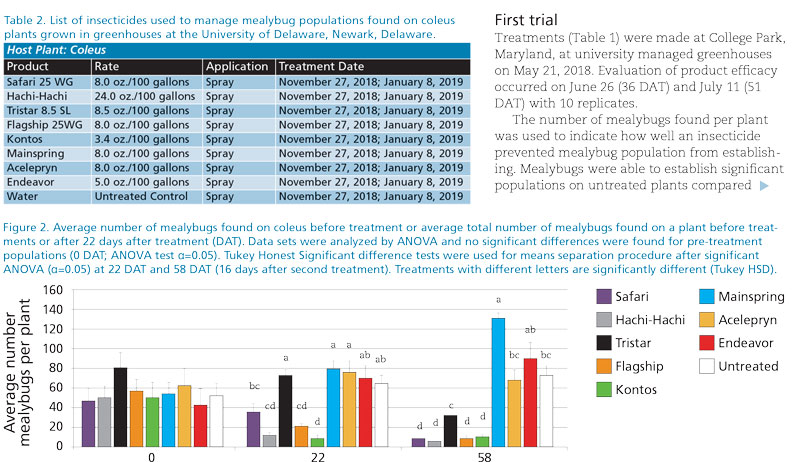
Second trial
A second trial was conducted in a greenhouse located at the University of Delaware on mealybug infested coleus. Pre-treatment counts were made November 27, 2018 and product evaluation was conducted 22 and 58 DAT (December 19 and January 24, 2019). This trial evaluated a variety of products against existing populations of citrus mealybug (Table 2) and had 10 replicates.
The total number of mealybugs found was used to determine insecticide efficacy. Prior to treatment, the coleus used in the experiment didn’t differ in the average number of citrus mealybugs found on each plant (F=0.96; df=8,72; P=0.4668; Figure 2). Hachi-Hachi, Flagship and Kontos all significantly reduced the average number of citrus mealybugs found on infested coleus 22 DAT (F=18.3; df=8,72; P<0.0001; Figure 2).
A second application of the insecticides in Table 2 was made on January 8, 2019 and the remaining mealybug population was counted on January 24. Safari, Hachi-Hachi, Flagship and Kontos significantly reduced mealybug populations compared to untreated controls (F=32.2; df=8,72; P<0.0001; Figure 2).
Summary
Citrus mealybug is one tough insect to control in the greenhouse environment and this is well-established with previous field research. When Altus, Ventigra, Acelepyrn, Mainspring and Transtect is applied to clean plants and infestation is moved into the growing area, we were able to keep the citrus mealybug at very acceptable levels for up to 51 days after treatment.
Many of the new chemistries tend to work preventatively as opposed to clean-up action, as demonstrated in our trials. Insect growth regulators (IGR) generally impact insects over 10 to 30 days; it may be similar with some of these new systemic insecticides. With many older insecticides acting as nerve poisons, they kill insects rapidly, but are more risk-oriented for the applicator. It’s important when using new chemistry that an IPM scouting program detects the pest early and treatment made before insect populations build up. Scouting pest-prone plants is essential with early interdiction. GT
Stanton Gill is Extension Specialist in IPM for Greenhouses and Nursery for the Central Maryland Research & Education Center, and University of Maryland Extension and Professor at Montgomery College. He can be reached at Sgill@umd.edu or (410) 868-9400. Brian Kunkel is an Extension Ornamental IPM Specialist for the University of Delaware Cooperative Extension. He can be reached at bakunkel@udel.edu or (302) 893-6077. Suzanne Klick is Lead Technician at the Central Maryland Research and Education Center for the University of Maryland. Rachel Ross is a Student Intern at the University of Maryland.