4/1/2019
Root Mealybug: What You Don’t See is Hurting Your Plants
Stanton Gill, Brian Kunkel, Chuck Schuster, Suzanne Klick & Rachel Ross

It’s human nature to ignore what you don’t see. Why worry about something if you can’t see it? As growers, you may notice the plant is stunted and not growing well. Until you take the plant out of the pot and look at the root system, then the wax associated with root mealybug becomes noticeable. If the population builds up, your customer will definitely notice a problem when they go to take the pot off and plant the infested plant—the wax is very noticeable on the roots on the outside perimeter of the rootball.
Pictured: Root damage from ground mealybugs, which also includes a small amount of wax that they secrete, giving the soil a somewhat bluish appearance when there’s a large population.
Three major insects groups can be problems on roots of nursery and greenhouse plants: fungus gnat larvae, root aphids and root mealybugs. We decided to concentrate on one of these pests in our trials and selected root mealybug. We selected this pest because it’s tough to control and mainly because we had a good population of the pest to work with in a nursery setting.
Root mealybug background
There are two major root mealybugs found on nursery and landscape plants in North America. One is called ground mealybug (Rhizoecus falcifera Kunckel d’Hercularis); the second is called Pritchard’s mealybug (Rhizoecus pritchardi McKenzie). Both are in the order Hemiptera, family Psuedococcidae.
The ground mealybug is white and 2.4 to 3.9 millimeters long, and has slender, waxy filaments that form a sort of netting over some individuals. The ground mealybug also secretes a small amount of wax, which can give the soil a somewhat bluish appearance when the mealybugs are abundant. Pritchard’s mealybug is smaller than the ground mealybug, and is snow white and 1.6 to 2.1 millimeters long and oval with very small eyes. The wax from Pritchard’s mealybug tends to be snow white in color. In our study, the Pritchard mealybug was present on sedum roots.
 The ground mealybug is reported to feed on the roots of anemone, chrysanthemum, gladiolus, iris and numerous other flowers, shrubs and ornamental grasses. Pritchard’s mealybug is a pest of achillea, arctostaphylos, aster, geum, polygala and sedum.
The ground mealybug is reported to feed on the roots of anemone, chrysanthemum, gladiolus, iris and numerous other flowers, shrubs and ornamental grasses. Pritchard’s mealybug is a pest of achillea, arctostaphylos, aster, geum, polygala and sedum.
Very little is published on the lifecycle of either root mealybug species. Dr. Arnold Hara of the University of Hawaii-Manoa reports female mealybugs lay eggs or give birth to live young called crawlers. If eggs are laid, they usually hatch in less than 24 hours.
Pictured: A green roof planting.
Crawlers are the dispersal stage and are highly mobile. Once the crawlers find a suitable site, they settle down and begin to feed on roots with their sucking mouth parts. The entire life cycle ranges from two to four months, depending on the species. Adults live from 27 to 57 days, also depending on the species.
Pritchard’s mealybugs have been noted to crawl out of drainage holes and spread throughout the nursery or greenhouse. In nurseries, we’ve found Pritchard’s mealybugs moving between pots of asters that were sitting on spun-down polypropylene weed barrier. The mealybugs were exiting through the drainage holes of the pot and moving from pot to pot that were growing close together. In green roof plant nurseries, we noted the Pritchard’s mealybugs moving from plug trays sitting on weed barrier on the ground to previously uninfested plant trays close to infested plants.
 Sedum + green roofs = root mealybugs
Sedum + green roofs = root mealybugs
Green roofs got their early start in Europe, mainly in Germany, as way to reduce water runoff from roof surfaces. We saw our first green roof in 2002 while visiting the Morton Arboretum in Lisle, Illinois, to speak on the newly emerging invasive emerald ash borer. While speaking there, they gave us an opportunity to visit some of the green roofs that had been installed in Chicago. The mayor then was very supportive of the green roof use in city roofs. Being our first time seeing this, we were very wonderstruck with this whole concept. It looked like a perfect opportunity for the horticulture industry to expand its scope.
Since then a number of cities have encouraged installation of green roofs to reduce energy use and clean up roof runoff water in city environments. Of course, there’s the aesthetic aspect that goes along with green roofs. Now people are adding benefits to pollinators and birds with green roof installation; the number of nurseries growing plants to be installed in these green roofs is growing.
Whenever anything is grown in large quantities in concentrated areas, you can guarantee that insects and disease will make their way into the system. This has happened with green roof plant production. Sedum plants have become the predominant group of plants that thrive on green roof plantings, so the nursery industry has concentrated on propagating large numbers of these succulent plants. At first, they appeared pest-free.
 Non-chemical control
Non-chemical control
Research conducted by the University of Hawaii College of Tropical Agriculture and Human Resources has demonstrated that hot water dips were effective in dealing with root mealybug in Hawaii. Submerging potted Rhapis palms in 120F (49C) water until the internal root ball temperature reaches 115F (46C) is 100% effective in killing root mealybugs and doesn’t significantly affect the potted plants.
In 2014, Stanton Gill of the University of Maryland Extension conducted trials on sedum; submersing the roots in 120F water for 15 minutes gave 100% control of root mealybugs. After the hot water treatments, the plants were cooled down to 60 to 70F (16 to 21C) for 10 minutes and returned to the growing area. The plants were examined at 14 and 28 days DAT and found to be undamaged by the hot water treatment. The method was effective, but slow and cumbersome, to treat large numbers of plug trays of sedum. For small numbers of plants, this may be a practical method.
Trials
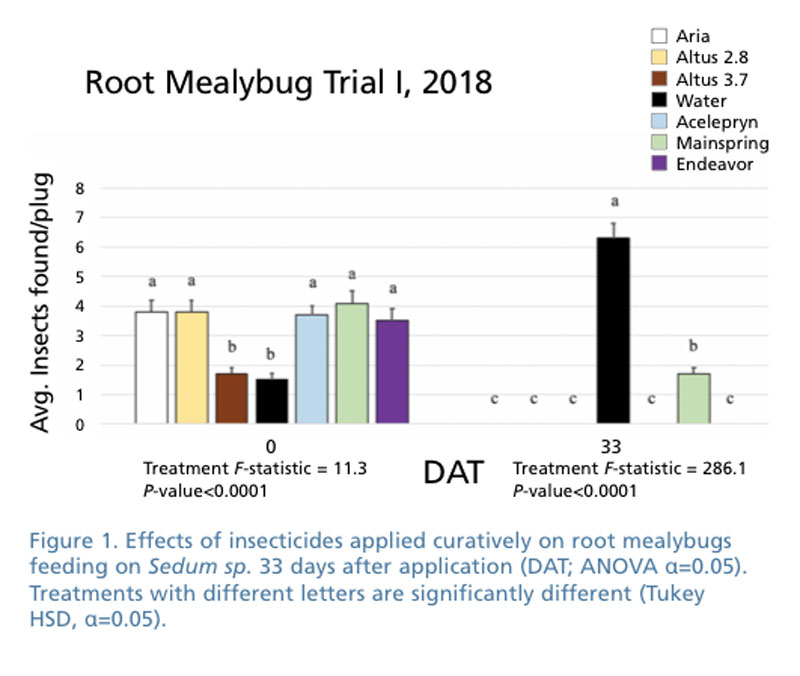
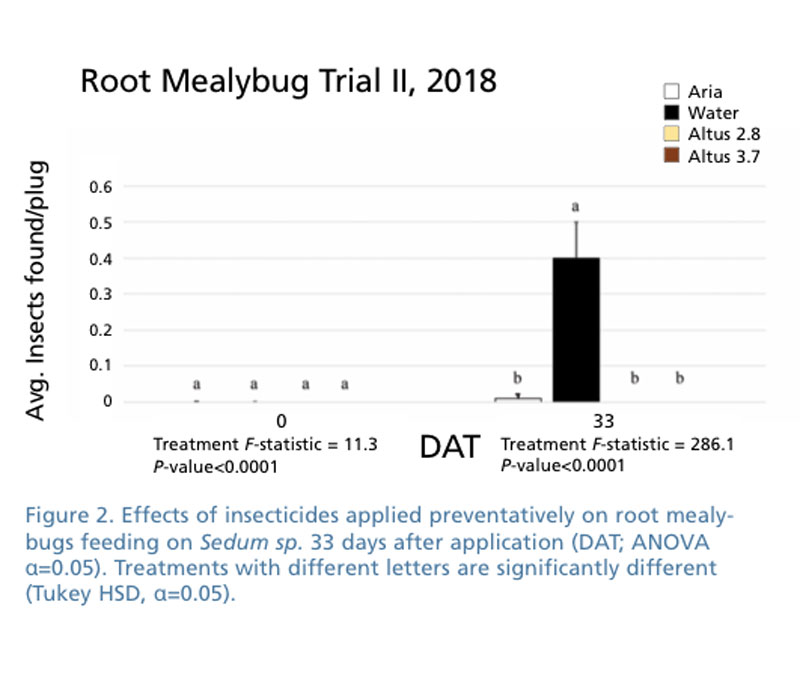
We worked with a green roof plant nursery that had sedums that were infested with both root aphid and root mealybugs. For this study, we concentrated on the root mealybug (Pritchard’s mealybug) in evaluating several new classes of systemic chemicals applied as soil drenches. Applications were made to clean plants with infested plug trays placed close to see if we could protect uninfested plants from becoming infested.
In the second trial, we used sedum plants in plug trays that were moderately to heavily infested with root mealybug to measure the control of infested plants. In each trial, we had eight completely randomized blocks. Treatments were made on April 5 or May 1, 2018 with each application being made as a soil drench.
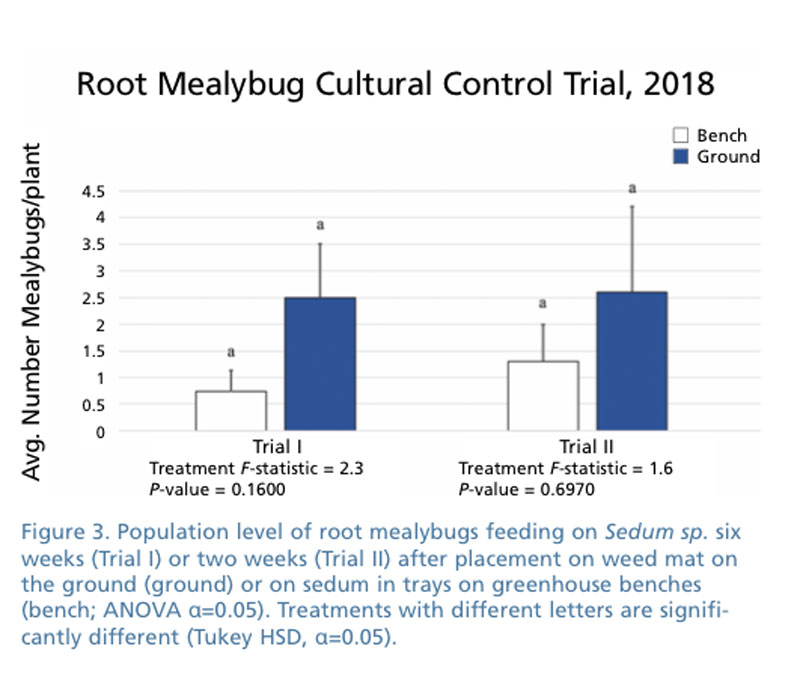 Our last trial against root mealybugs focused on where plants were grown. Our treatments were growing sedum in flats on weed mat in contact with the soil (served as the industry standard) and plants grown in flats on greenhouse benches placed in a hoophouse next to the other plants. Populations were similar prior to beginning the experiment. Two population counts were made on plants grown in the hoophouse, as described previously after six (Trial I) and two (Trial II) weeks after starting the trial.
Our last trial against root mealybugs focused on where plants were grown. Our treatments were growing sedum in flats on weed mat in contact with the soil (served as the industry standard) and plants grown in flats on greenhouse benches placed in a hoophouse next to the other plants. Populations were similar prior to beginning the experiment. Two population counts were made on plants grown in the hoophouse, as described previously after six (Trial I) and two (Trial II) weeks after starting the trial.
Summary
All of the treatments were effective in keeping root mealybug from spreading from infested flats to uninfested flats. In the first trial involving treating already infested sedum that had established root mealybug, we found Aria, Altus, Acelepryn, Endeavor and Mainspring provided effective control, although populations were slightly higher on Mainspring-treated plants (Figure 1). Altus and Aria were effective preventative treatments (Figure 2).
We noted during our trials that when we took flats from a nursery growing the flats on black weed barrier and moved them into a greenhouse on wire bench the root mealybugs population tends to decline rather dramatically (Figure 3.). It may be the growing method of placing plastic flats on the ground that allow root mealybug and root aphids to easily travel from infested flats to previously uninfested flats. The two different locations to grow plants weren’t significantly different in either trial. Possible reasons for no significant differences include length of our experiments (two or six weeks) or the number of plants evaluated. We plan to investigate this further with future trials.
 Hot-water treatments of 120F (49C) for 15 minutes appear to control root mealybug, but the method is slow, slightly cumbersome, time-consuming and good for treating already infested plants. Preventative insecticide treatments appear to keep plant free of infestation of root mealybug. GT
Hot-water treatments of 120F (49C) for 15 minutes appear to control root mealybug, but the method is slow, slightly cumbersome, time-consuming and good for treating already infested plants. Preventative insecticide treatments appear to keep plant free of infestation of root mealybug. GT
Stanton Gill is Extension Specialist in IPM for Greenhouses and Nursery for the Central Maryland Research & Education Center, and University of Maryland Extension and Professor at Montgomery College. He can be reached at Sgill@umd.edu or (410) 868-9400. Brian Kunkel is an Extension Ornamental IPM Specialist for the University of Delaware Cooperative Extension. He can be reached at bakunkel@udel.edu or (302) 893-6077. Chuck Schuster is an Extension Educator at the University of Maryland Extension. He can be reached at CFS@umd.edu. Suzanne Klick is Lead Technician at the Central Maryland Research and Education Center for the University of Maryland. Rachel Ross is a Student Intern at the University of Maryland.