7/1/2018
Dipping with Biopesticides
Michael Brownbridge
“To dip or not to dip? That is the question.”
I’m not sure if Hamlet ever grew flowers, or actually said that, but what we do know is that dipping cuttings in biopesticides is a great way to get a head start on a biocontrol program. Here’s why …
Many floriculture crops are started from vegetative cuttings to maintain the stability of desired traits (cuttings essentially produce clones of the parent) and ensure a high success rate in propagation.
Greenhouses in Ontario, for example, import cuttings from propagators based in South and Central America, East Africa, New Zealand, Southeast Asia, Europe and the U.S., and propagate other floriculture crops from cuttings taken in-house. While propagators take exceptional measures to keep pests under control, cuttings frequently come with unwanted hitchhikers. These “starter cultures” of pests are very hard to detect in batches of thousands of cuttings. Numbers are often very low, and in the case of insects like Western flower thrips, eggs are deposited in plant tissue and impossible to see from a superficial examination of incoming cuttings. Left untreated, a few can become problematic very quickly.
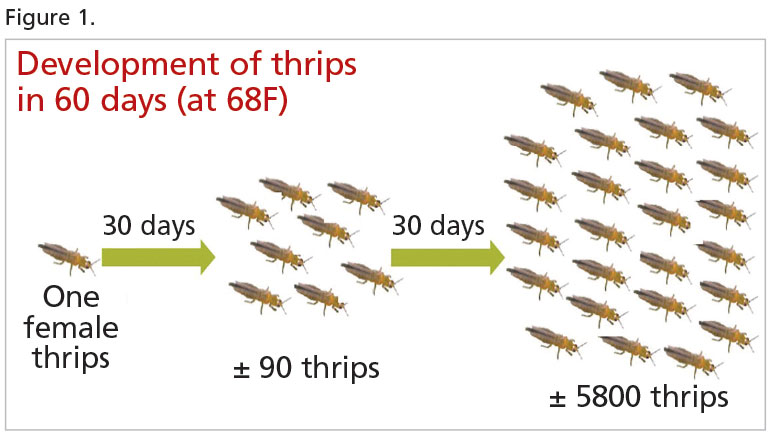 For example, one female Western flower thrips can potentially generate a population of over 5,500 in just 60 days (Figure 1) and Bemisia whiteflies can go from two to over 5,000 in 42 days. Clearly, the sooner you can intervene and nip these populations in the bud (no pun intended!), the better.
For example, one female Western flower thrips can potentially generate a population of over 5,500 in just 60 days (Figure 1) and Bemisia whiteflies can go from two to over 5,000 in 42 days. Clearly, the sooner you can intervene and nip these populations in the bud (no pun intended!), the better.
Figure 1 Graphic reproduced with the kind permission of Ronald Valentin, Bioline AgroSciences.
But there are more twists in this plot. The global trade in propagative material explains, in part, why some pests have global status; it’s also a way in which resistance can spread. Pest management programs at most propagators rely heavily on the use of insecticides. There’s a high likelihood that pests carried on cuttings will already have been exposed at some point to materials that are registered in the U.S. and Canada. As a result, pests may already be resistant to some of these insecticides, while the activity of others may be compromised.
Also, cuttings often carry pesticide residues, which can impact the success of a (preventative) biocontrol strategy. Residues may not be directly toxic, but sub-lethal effects can reduce the fitness of natural enemies, diminishing their efficacy and capacity to establish, and shorten their life span. So what immediate steps can be taken to take care of these incoming pests?

Take them for an early dip
Low-risk materials and biopesticides can play an important role. Dipping cuttings (and plugs) in biopesticides is a particularly efficient way of eliminating pests on propagative material at a time when it’s difficult to obtain good spray coverage. Immersion of cuttings provides excellent coverage of the foliage and a large number of cuttings can be quickly and effectively treated at the same time, using a relatively small amount of product (Figure 2).
It’s a technique that can be readily integrated into the general work flow, a step done ahead of sticking for example, and the use of biopesticides minimizes risks to workers. (Note: The process still requires the use of protective gear.) Also, biopesticides have short residuals, meaning that biocontrol agents can be released almost immediately after treatment of the cuttings.
The biopesticide dip doesn’t eradicate all of the pests on the material, but it will eliminate a significant proportion, effectively slowing population growth so that biocontrol strategies have a higher likelihood of success. Consequently, this early intervention technique strengthens biologically-based IPM programs, resulting in more robust, efficacious and cost-effective systems.
The technique is very effective in poinsettias, and in Ontario, incoming cuttings are routinely dipped in a combination of BotaniGard WP and insecticidal soap to control Bemisia whiteflies. (This short video demonstrates best practices: www.youtube.com/watch?v=qF5bjLriaTg.) We found that this step eliminated around 70% of the whiteflies on infested leaves, which had a major impact on population growth thereafter.
Dips are followed by a regular biocontrol program, which incorporates parasitoids (Encarsia, Eretmocerus), predatory mites (Swirskii) and occasional sprays with BotaniGard. Following the use of this technique, synthetic pesticides are often not required, or at most, their use is limited to one or two clean-up sprays towards the end of the production cycle.
Research underpinning the development of these dipping protocols not only considered product efficacy, but also investigated potential phytotoxicity (product, rate and poinsettia variety) and phytosanitary risks. The work validated product efficacy and defined safe or only mildly phytotoxic rates to some varieties. To address the threat of disease transfer, we considered the highly contagious plant pathogen Pectobacterium carotovora (Erwinia). Although low levels of Pectobacterium were generally present in dip suspensions used in commercial operations, no disease symptoms were observed in crops.
Furthermore, in experimental trials where the dip suspension was artificially “spiked” with Pectobacterium, the disease was only seen when extremely high concentrations of the pathogen were present. We concluded that the risks of disease transfer are very low, but it’s good practice to periodically change the suspension and sanitize all equipment regularly to avoid the build-up of pathogens to infectious levels.
Of course, dipping is not a panacea; we see it as an integral part of a larger program where a “systems approach” is taken to pest management, which combines biological control and plant resistance with the manipulation of environmental and crop production practices to suppress pest populations. In many cases, dipping may be an enabling step that facilitates the successful use of biological control tactics thereafter—part of a “clean start” approach that’s the foundation of any IPM strategy (Figure 3).
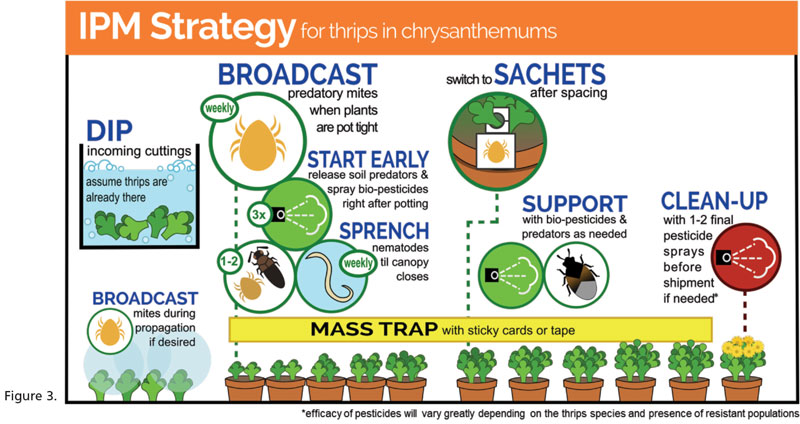
Figure 3 Info-graphic developed by Ashley Summerfield, Vineland Research and Innovation Centre. Not to be copied without permission. See GreenhouseIPM.org for more details.
The simplicity of the method makes it an attractive option for other propagative crops and pests. Could the technique be used to manage thrips or spider mites on chrysanthemum or mini-rose cuttings? Do the same products and rates that work well on poinsettia do a similar job on these crops/pests? Can the approach be readily integrated with other components of a larger crop management program and support the targeted outcome of more sustainable plant production? That’s why we do research! GT
Acknowledgements: The author would like to thank researchers and technical staff who contributed to this study: Dr. Rose Buitenhuis, Dr. Sarah Jandricic, Graeme Murphy, Taro Saito, Ashley Summerfield, Wonhyu Lee, Mark Jandricic, Angela Brommit, Paul Côté, Rebecca Eerkes and Erfan Vafaie. The project on Bemisia whitefly management for poinsettia was funded in part through Growing Forward 2 (GF2), a federal-provincial-territorial initiative. The Agricultural Adaptation Council assisted in the delivery of GF2 in Ontario. Additional support was provided by Dümmen Orange, BioWorks Inc., Koppert Canada and Flowers Canada (Ontario) Inc. (Guelph, Ontario, Canada).
Michael Brownbridge is the Research Director, Horticultural Production Systems at Vineland Research and Innovation Centre in Ontario, Canada.