8/31/2017
Understanding pH Personality
Ryan Dickson & Paul Fisher
Even though we tend to grow many crops with the same root substrate, fertilizer and lime, the “pH personality” of species can differ in two key ways: (1) susceptibility to micronutrient toxicity at low pH or deficiency at high pH; and (2) tendency to make pH drift up (more basic) or down (more acidic).
Substrate-pH influences nutrient solubility and availability for root uptake, especially iron and other micronutrients such as manganese and boron. Plants only take up nutrients that are soluble and dissolved in water, and most micronutrients decrease in solubility as pH increases. For example, seed geranium is susceptible to iron toxicity at low pH (Figure 1A). In contrast, petunia is susceptible to iron deficiency at high pH (Figure 1B).
 Figure 1. Iron toxicity symptoms in lower leaves of geranium (A). Iron deficiency symptoms in upper leaves of petunia (B).
Figure 1. Iron toxicity symptoms in lower leaves of geranium (A). Iron deficiency symptoms in upper leaves of petunia (B).
Some crops have a natural tendency to run into trouble. These are the canaries in the coal mine or “bioindicator” species that are most likely to show pH is too low or high. Geraniums tend to push pH down during production, even though they’re sensitive to iron toxicity at low pH. Petunias tend to push pH up, although they’re susceptible to iron deficiency.
However, our research has found that a crop’s sensitivity to high or low pH is often not related to their tendency to change pH over time. Understanding these concepts will help you design a preventative fertilizer program that avoids pH issues.
pH Sensitivity: Stress at low or high pH
Plant species differ in how efficiently they take up micronutrients, which influences how susceptible they are to toxicity or deficiency. We can group floriculture crops into three pH sensitivity categories:
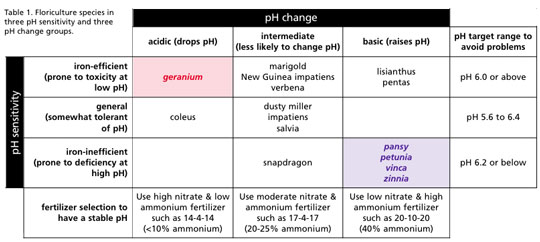
• Iron-efficient species: These species are very efficient at taking up micronutrients and tend to be susceptible to iron toxicity, especially at low pH. Examples of iron-efficient crops are geranium, marigold, lisianthus, New Guinea impatiens, pentas and broad-leafed verbena. These plants should be grown at a high pH 6.0 to 6.6.
• Iron-inefficient species: These species are inefficient at taking up micronutrients and tend to be susceptible to iron deficiency, especially at high pH. Examples of iron-inefficient crops are petunia, pansy, zinnia, snapdragon, vinca and calibrachoa. These plants should be grown at a low pH of 5.4 to 6.2.
• General species: These species are less susceptible to developing micronutrient disorders and can usually tolerate a wider range in pH. Examples of general crop species are impatiens, coleus, salvia, dusty miller, garden verbena and chrysanthemum. These plants can usually tolerate pH 5.6 to 6.4.
pH Change: Tendency to change substrate pH
We conducted research at the University of Florida to evaluate the tendency of floriculture species to push pH up or down. Fifteen crop species were irrigated with three fertilizer formulations ranging from 0:100, 20:80 or 40:60 ammonium:nitrate nitrogen (N) ratio. Plants were grown with deionized water (no alkalinity). The 70% peat/30% perlite substrate was mixed pre-plant using hydrated lime, which didn’t provide residual buffering to pH change.
Ammonium N tends to be strongly acidic and nitrate N tends to be slightly basic. Fertilizing with 100% nitrate (no ammonium) caused plants in our trial to raise substrate pH, except for geranium, which had a stable pH (no pH change). Increasing ammonium up to 40% of fertilizer nitrogen (60% nitrate) caused all species to produce acid and lower pH.
Species were statistically separated into three pH change groups termed “acidic” (drop pH over time), “intermediate” (little pH change) and “basic” (raise pH).
Species in the “acidic” pH change group (coleus and geranium) had a neutral pH effect (no pH change) when fertilized with 0% ammonium (100% nitrate) and were strongly acidic when ammonium increased to 40% (60% nitrate). Species in the “basic” pH change group (lisianthus, pansy, pentas, petunia, vinca and zinnia) were strongly basic with 0% ammonium and weakly acidic with 40% ammonium. Remaining “intermediate” crops (dusty miller, impatiens, marigold, New Guinea impatiens, salvia, snapdragon and verbena) caused a pH reaction that was intermediate between the acidic and basic species.
How does this affect crop management?
Table 1 summarizes where the 15 species we tested lie within pH sensitivity and pH change groups. Without wanting to make fertilizer management too complex, appreciate that crop species have their own “pH personality” that differs in terms of pH sensitivity and pH change. Many crops are unlikely to run into pH issues—they’re either less sensitive to pH range or they tend to change pH in the “right direction” (toward their target pH range).
However, one problem pH personality type is represented by geranium (sensitive to micronutrient toxicity at low pH, but also pushes pH down). With these plants, consider adding additional lime to start with a higher pH and provide buffering to a drop in pH. Do not apply excess ammonium fertilizer to these plants.
A second problem pH personality is represented by pansy, petunia, vinca and zinnia. These plants are sensitive to high pH and with their basic pH change will push themselves into iron deficiency problems. Use an acidic reaction (high ammonium fertilizer), consider reducing pre-plant lime rate, and if water alkalinity is high, then acidification of the water supply may be needed.
Keys to success in pH management involve understanding the pH personality of your crops species to identify the “usual suspects” of high or low pH. In addition, the correct fertilizer and lime selection, and regular testing of substrate pH and electrical conductivity will keep crops in the target pH range.
GT
Acknowledgements: Thanks to funding support and collaboration from industry partners of the Floriculture Research Alliance (www.floriculturealliance.org) as well as the USDA Floriculture and Nursery Research Initiative, and the Gene and Barbara Batson Scholarship.
Ryan Dickson is an Associate Extension Professor at the University of New Hampshire (ryan.dickson@unh.edu) Paul Fisher is a Professor and Extension Specialist at the University of Florida (pfisher@ufl.edu).