7/31/2017
Tips for Rooting Perennial Cuttings, Part 2
W. Garrett Owen & Roberto G. Lopez
Adventitious root formation and development (Stage 3) of vegetatively propagated perennials is a complex process that’s influenced by exogenous and endogenous factors. While propagators cannot control endogenous factors (genetics, plant hormones, mineral nutrition and carbohydrate status of cuttings), they can control exogenous factors (air and root-zone temperatures, mist irrigation and moisture, fertility, humidity, and light).
Though environmental conditions can be manipulated during root development, optimizing these conditions is often challenging since herbaceous perennials are rooted year-round under varying propagation daily light integrals (PDLI) and temperatures (air and root-zone). Additionally, callusing and rooting cuttings quickly and evenly with more than 20 genera of perennials in one propagation area is a challenge often expressed among
propagators.
Therefore, our ongoing research at Michigan State University is quantifying how PDLI, RZT and air temperature interact and can be optimized during herbaceous perennial callusing and rooting for a wide range of genera. In this last article of a two-part series, we’ll highlight our propagation photosynthetic lighting and root-zone temperature research on adventitious root formation and development.
Rooting experiment
Unrooted shoot-tip herbaceous perennial cuttings of
Achillea millefolium Apricot Delight (common yarrow), Agastache hybrid Purple Haze (anise hyssop),
Coreopsis rosea Limerock Ruby (tickseed),
Gaillardia aristata Gallo Red (blanketflower),
Gaura lindheimeri Siskiyou Pink (wandflower),
Heuchera hybrida Black Beauty (coral bells),
Lamium maculatum Purple Dragon (spotted deadnettle),
Leucanthemum x superbum Snowcap (shasta daisy),
Perovskia atriplicifolia (Russian sage),
Phlox paniculata Bright Eyes (garden phlox), and
Salvia nemerosa Blue Hill (meadow sage) were received from commercial cutting suppliers. Cuttings were stuck in 72-hexagon cell liner trays without rooting hormone. The trays were filled with a commercial soilless substrate amended with 50% coarse perlite. The cuttings were subsequently sprayed to runoff with a surfactant to ensure a uniform coverage of moisture on the leaves during
propagation.
Trays were then placed in a glass-glazed propagation greenhouse to be callused under a DLI of ≈5 mol•m–2•d–1, air temperature of 73F (22C), RZT of 74F (23C), and relative humidity of 80%. These set points were chosen based on results from the callusing experiment discussed in the first article (
GrowerTalks, July 2017) of the series. After three, five or eight days (genera specific), cuttings were transferred into another glass-glazed propagation greenhouse for rooting.
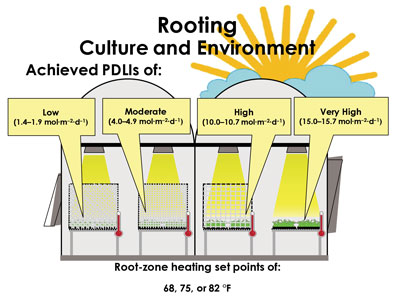
In the rooting greenhouse (Figure 1), cuttings were placed under ≈86, 62, 26% or 0% shade with supplemental lighting delivered from high-pressure sodium lamps to provide a 16-hour photoperiod. As a result, we created four PDLI treatments: low (1.4 to 1.9 mol•m–2•d–1), moderate (4.0 to 4.9 mol•m–2•d–1), high (10.0 to 10.7 mol•m–2•d–1) and very high (15.0 to 15. 7 mol•m–2•d–1).
Figure 1. Callused cuttings were transferred to a greenhouse where they were rooted under ≈86, 62, 26% or 0% shade with supplemental lighting from HPS lamps for 16 hours to achieve four propagation daily light integral (PDLI) treatments: low (1.4 to 1.9
mol•m–2•d–1), moderate (4.0 to 4.9
mol•m–2•d–1), high (10.0 to 10.7 mol•m–2•d–1) and very high (15.0 to 15. 7 mol•m–2•d–1). Under each PDLI treatment, medium temperatures were maintained with root-zone temperature (RZT) set points of 68, 75 or 82F.
Under each PDLI treatment, a root-zone heating system circulated hot water across the bench top of each expanded metal propagation bench. The benches were insulated with foam board to reduce heat loss and covered with galvanized sheet metal that evenly disbursed heat across the bench top. Each propagation bench was programmed and controlled individually to maintain RZT set points of 68, 75 or 82F (20, 23 or 27C). Cuttings were rooted under air and relative humidity set points of 70F (21C) and 60%, respectively. Mist consisting of reverse-osmosis water was controlled and applied beginning and ending one hour before and after the photoperiod and was adjusted for each PDLI and RZT treatment. After eight days, mist was discontinued and cuttings were over-head irrigated once daily with reverse-osmosis water supplemented with a water-soluble fertilizer and micronutrient supplement providing 60 ppm nitrogen.
Results
For all herbaceous perennials in our study, with the exception of coral bells, we found cutting quality and root development to be poor under low (1.4 to 1.9 mol•m–2•d–1) and moderate (4.0 to 4.9 mol•m–2•d–1) PDLIs at a RZT of 68F (20C). Cutting quality improved as RZT increased from 68F to 75 and 82F, but we found the minimum acceptable PDLI to be 4.0 to 4.9 mol•m–2•d–1 (moderate). Even though warmer RZTs improved cutting quality under moderate PDLIs, it did not result in the greatest cutting quality (large root systems and thick stems) that was achieved under higher PDLIs.
After 14 days in the rooting environment, blanketflower root mass increased and cuttings were studier (thick stem caliper) and more compact (shorter stems) when a RZT of 75F and a high PDLI was maintained (Figure 2). Blanketflower growth and development occurred faster under high and very high PDLIs and RZTs of 75 and 82F and we observed more flower bud initiation and formation because plant development is temperature-dependent.
Wand flower (Figure 3) and tickseed (Figure 4) cutting quality was greatest under high and very high PDLIs at each RZT. We also found wand flowers developed faster under higher PDLIs and at RZTs of 68, 75 and 82F. Though cuttings were often taller at higher RZTs, it didn’t diminish cutting quality compared to those propagated under lower PDLIs. Unlike the 10 other herbaceous perennials, coral bells rooted best under moderate and high PDLIs at RZT set points of 68 and 75F (Figure 5). We did observe coral bell cuttings to produce little to no roots at RZT of 82F, therefore, we do not recommend using RZT >75F when propagating coral bells.
Results of these experiments determined species-specific responses to increasing PDLIs at 68, 75 and 82F RZTs throughout rooting. Our research provides evidence that providing a PDLI of 10 mol•m–2•d–1 and RZT of 75F during root development (Stage 3) when propagating a wide range of unrooted perennial cuttings can increase cutting growth, development and overall finished rooted cutting quality and reduce rooting time.
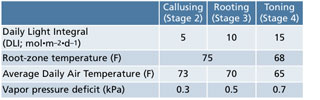 Table 1. General recommended daily light integral, root-zone temperature, air temperature and vapor pressure deficit for callusing, rooting and toning perennial cuttings (Coral bells is the exception).
Table 1. General recommended daily light integral, root-zone temperature, air temperature and vapor pressure deficit for callusing, rooting and toning perennial cuttings (Coral bells is the exception).
When the results of several studies are combined for the 11 genera and cultivars we’ve investigated thus far, we recommend following the DLI, RZT, air temperature and vapor pressure deficit guidelines in Table 1 for most genera with the exception of coral bells. We hypothesize that much of the increased growth and development may be attributed to elevated photosynthesis and plant temperature for cuttings propagated under increasing PDLIs and RZT, respectively.
Additionally, the reduced propagation time allows producers to increase the number of crop turns in their production facility and save on energy costs. Furthermore, our results on the influence of PDLI and RZT during propagation will enable producers to make better informed decisions for managing supplemental lighting and RZT throughout callusing and rooting.
GT
Thanks to a Michigan State University Project GREEEN grant (www.canr.msu.edu/research/plant-agriculture/project_greeen/) and continued Fred C. Gloeckner Foundation (www.gloecknerfoundation.org/) funding, we’ll now expand our perennial propagation research to include light quality and reduced air temperatures.
W. Garrett Owen (wgowen@msu.edu) is a Floriculture Outreach Specialist and Roberto Lopez (rglopez@msu.edu) is an Assistant Professor and Extension Specialist in Controlled Environment and Floriculture Production at Michigan State University. They gratefully acknowledge Rob Eddy, Kyra Brown, Hilary Burkhardt, Joshua Craver, Melissa Grant, Jun Hayashi, Andrea Hilligoss, Wyatt Lucas, Jill Malecki, Kyle Martin, David Myers, Madeline Olberg, Zach Oravec, and Brenton Strim for greenhouse and laboratory assistance. They would like to thank Ball Horticultural Co. and Dümmen Orange for plant material; Sun Gro Horticulture for substrate; Ludvig Svensson for shade cloth; J.R. Peters, Inc. for fertilizer; and Michigan State University Project GREEEN grant, the Fred C. Gloeckner Foundation, Inc. and the New Hampshire Plant Growers Association for financial support.