5/31/2017
Chemistry Crash Course
Ryan Dickson & Paul Fisher

Seed and zonal geraniums often push substrate pH down in the growing medium, which can result in iron and manganese toxicity symptoms. A low-substrate pH increases solubility and uptake of iron and manganese by plant roots. Plants take up so much of these micronutrients that yellowing occurs, cells die in old leaves and crop losses occur (see Figure 1). But why does this happen?
Figure 1. Lower leaf yellowing and necrosis in geranium caused by iron/manganese toxicity at low pH.
We explain here why geraniums add acid into the root zone and drop substrate-pH, even when the initial substrate-pH is above 6.0. By understanding this process, growers can take actions to prevent this problem.
Even when all crops receive the same fertilizer under equal growing conditions, different species can cause substrate-pH to drift over time. For example, over a series of trials, we’ve seen that geraniums will consistently tend to drop pH, petunias will tend to raise pH and bedding plant impatiens are intermediate.
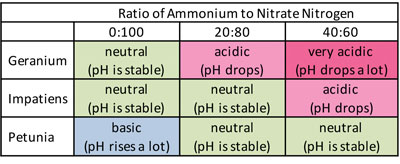 Table 1. pH effects of plant species grown with different fertilizer ammonium (acid) to nitrate (basic) ratios when using 100 ppm N in zero alkalinity water.
Table 1. pH effects of plant species grown with different fertilizer ammonium (acid) to nitrate (basic) ratios when using 100 ppm N in zero alkalinity water.
Table 1 summarizes our research trials for geraniums, impatiens and petunias grown with different nutrient solutions. In this scenario, the irrigation water is deionized water (zero alkalinity). The water-soluble fertilizer is applied with each irrigation at a concentration of 100 parts per million (ppm) of nitrogen and different ratios of ammonium to nitrate nitrogen. For example, an ammonium:nitrate ratio of 0:100 would contain no ammonium and all nitrogen in the nitrate form.
If we take the example of 40:60 (which is the same ammonium:nitrate ratio as in the common fertilizer 20-10-20), geranium grown with this fertilizer will drop pH a lot, impatiens will drop pH slightly and petunia will be neutral. So the ammonium:nitrate fertilizer with a neutral effect will differ depending on plant species.
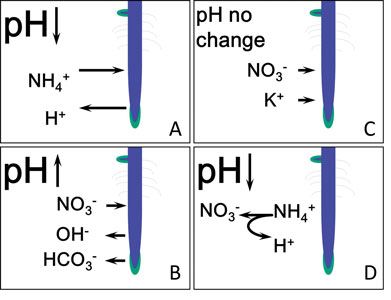 Figure 2. Effects on substrate-pH from fertilizer uptake by roots. (A) Cation uptake is acidic. (B) Anion uptake is basic. (C) When roots take equal cations and anions pH doesn’t change. (D) Nitrification of ammonium by microbes is acidic.
Figure 2. Effects on substrate-pH from fertilizer uptake by roots. (A) Cation uptake is acidic. (B) Anion uptake is basic. (C) When roots take equal cations and anions pH doesn’t change. (D) Nitrification of ammonium by microbes is acidic.
However, whether the species effect is enough to push substrate pH up or down over time in your crop also depends on other factors. Media components, such as peat and bark, are acidic (low pH around 3 to 5), coir and perlite are neutral, and vermiculite is basic (above pH 7). Ammonium nitrogen is acidic, especially when applied at high concentration, whereas nitrate is basic. Lime is added to media to increase substrate pH to around 6.0 and balance the acidity of peat or bark. Alkalinity of irrigation water is like dissolved lime, raising substrate pH with each watering.
Plant uptake of nutrients affects substrate pH
So why do geraniums drop pH more than petunia and why is ammonium more acidic than nitrate? A little chemistry is needed to understand how plant roots interact with nutrients to affect substrate pH.
Plants take up “cations,” which are nutrients with a positive charge, such as ammonium NH4+, potassium K+, calcium Ca2+ and magnesium Mg2+. Plants also take up “anions” with a negative charge, such as nitrate NO3-, phosphate H2PO4- and sulfate SO42-.
Taking up a cation such as NH4+ makes the plant more positively charged than the soil. To balance this charge difference between plant and soil, roots exude an acid (H+), which drops the substrate-pH (Figure 2A).
When plants take up an anion such as NO3-, the plant balances the charge by releasing a base into the soil (hydroxyl OH- or bicarbonate HCO3-), which increases substrate-pH (Figure 2B). Plants can also sometimes take up equal amounts of cations and anions, which results in no pH change (Figure 2C).
Nitrogen is especially important to substrate pH because: (a) Roots can take up nitrogen as a cation (ammonium NH4+) or anion (nitrate NO3-); and (b) plants require more nitrogen than any other fertilizer nutrient. Ammonium is a stronger acid than nitrate is a base, in part because nitrifying bacteria present in the growing medium also produce acid by converting ammonium nitrogen to nitrate through a process called nitrification (Figure 2D). Many plants also favor ammonium uptake over nitrate.
Species and fertilizer affect substrate pH
In our research, we’ve used geranium, impatiens and petunia as examples of species with acidic, neutral or basic effects on substrate pH, respectively. Plants were grown in both hydroponic nutrient solutions and peat substrates. We varied nitrogen form between ammonium (NH4+) and nitrate (NO3-), and tested substrate pH and the nutrients taken up by each species.
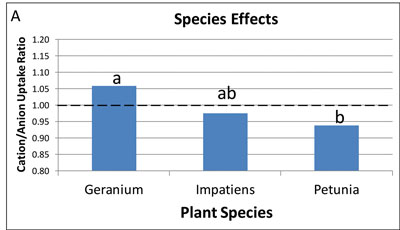 Figure 3. The ratio of plant uptake of positive cations, such as ammonium versus uptake of anions such as nitrate. (A) Differences between species. (B) Differences between ammonium:nitrate ratio in the fertilizer. Cation/anion uptake ratio values greater than 1 are acid, less than 1 is neutral and equal to 1 is basic.
Figure 3. The ratio of plant uptake of positive cations, such as ammonium versus uptake of anions such as nitrate. (A) Differences between species. (B) Differences between ammonium:nitrate ratio in the fertilizer. Cation/anion uptake ratio values greater than 1 are acid, less than 1 is neutral and equal to 1 is basic.
When grown with the same fertilizer, species differed in cation or anion nutrient uptake (Figure 3). Because geranium tends to favor uptake of cations such as ammonium (cation/anion uptake ratio > 1), it tends to exude acid resulting in a lower substrate pH. Petunia has the opposite trend, favoring anion uptake (cation/anion uptake ratio < 1) and pushing up substrate pH.
Increasing ammonium in the nutrient solution increased cation uptake and acidity produced for all three species (Figure 3B). Uptake of ammonium occurs more rapidly than nitrate for most plant species, increasing total ca
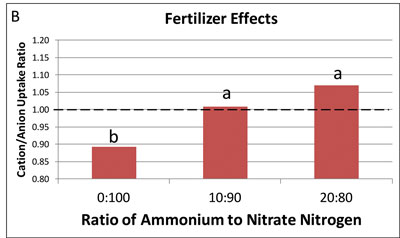
tions taken up and decreasing root zone pH.
Conclusions
A fairly typical scenario is that a greenhouse produces multiple bedding plant species with the same container substrate and an acidic fertilizer such as 20-10-20. The initial substrate pH is around 6.0. However, several weeks into the crop, the pH for geraniums drops, resulting in iron/manganese toxicity.
This typically occurs when plant roots are large relative to the container, the plant is taking up nutrients quickly and applied fertilizer concentration is increased. All these factors favor ammonium uptake. Geraniums take up the cations, such as ammonium, and exude acid. In addition, lime may have all reacted (been used up) in the substrate, resulting in less buffering to pH change. If fertilizer level is high, and it contains more than 20% of the nitrogen in the ammonium form, pH can drop by a point within one to two weeks.
Although we often treat crop species the same, they’ve evolved in different environments. Geraniums are “iron-efficient” and can take up micronutrients in naturally high-pH calcareous soils. They’ve evolved to take up more cations than anions, exude acid from roots and make iron more available to avoid deficiency. When we put these geraniums in a fertilizer-rich and poorly buffered container substrate, they still drop pH and run into problems. In contrast, iron-inefficient petunias have evolved in soil types richer in available micronutrients, resulting in the opposite behavior of raising pH and suffering iron deficiency in container substrates.
One method to handle differences between species discussed in this article is to vary the ammonium:nitrate ratio of the fertilizer. The species pH effect can be balanced with the fertilizer type. You could grow geraniums with a low-ammonium and high-nitrate fertilizer (a “basic” reaction fertilizer with less than 20% of N as ammonium and urea) to avoid a pH drop. Using a more acidic fertilizer (>30% of N as ammonium and urea) will help prevent an increasing pH and iron deficiency symptoms in petunia. However, varying fertilizer strategy isn’t always practical if you have only one nutrient injector and a future article (scheduled for the September issue) will cover several practical methods to deal with species differences in pH response.
GT
Acknowledgements. Thanks to funding support and collaboration from industry partners of the Floriculture Research Alliance (www.floriculturealliance.org) and USDA Floriculture and Nursery Research Initiative.
Ryan Dickson is an Associate Extension Professor at the University of New Hampshire (ryan.dickson@unh.edu). Paul Fisher is a Professor and Extension Specialist at the University of Florida (pfisher@ufl.edu).