3/29/2017
Take Your Poinsettia Cuttings for a Dip
Rose Buitenhuis

Where do pests come from? Sometimes they seem to just magically appear on plants. In reality, there are a few possible sources of pests: An existing infestation can go mostly undetected and pop up at the most inconvenient times when plants are susceptible or when treatments controlling them fail.
Pests can also come in from outside, through unscreened vents or doors. Often, infestations are related to certain periods of the year or weather events. Finally, despite the best efforts by propagators to deliver clean material, pests may be coming in on propagative plant material, like cuttings or plugs. They’re excellent hitch hikers and travel around the world anywhere plant material is shipped.
Figure 1. Cuttings are placed in a tray and immersed in a biopesticide solution.
Low levels of pests on cuttings are very hard to detect and their presence or absence can make or break the success of a pest management program. Pest populations inevitably increase rapidly, and once they reach a certain level, biocontrol agents cannot catch up. Intervention with one or more pesticides may be needed, potentially disrupting biocontrol programs. Even if you’re using a chemical pest management approach, these pests may be difficult to kill because of their resistance as a result of repeated exposure to the same products at the propagation facility.
So what to do? It’s important to scout incoming plant material. Still, dealing with pest life stages that are (nearly) invisible, such as eggs, and a large number of boxes of plant material to examine in a short time when cuttings are received, it may be like looking for a needle in a haystack. For many plant species, it’s safe to assume that incoming material will be infested with one or more pests, like thrips, whiteflies or spider mites. Thus, the earlier you can do something about it, the better.
Take a dip
One technique that allows growers to “start clean” are cutting dips. When cuttings are unpacked from the shipping boxes, they’re spread out loosely in a mesh tray, covered with a second tray and immersed in a biopesticide solution (Figure 1). Following dipping, cuttings are planted as usual. This technique is fast, easy to integrate into the workflow and ensures complete coverage of cuttings with the biopesticide. The advantage of using biopesticides is that they have no or a short residual effect and are highly compatible with a biocontrol program.
To demonstrate the benefits of cutting dips, we did a three-year case study on poinsettias at Vineland Research and Innovation Centre (Ontario, Canada). One of the main pests on poinsettia is the whitefly, Bemisia tabaci. Since this whitefly doesn’t survive Canadian winters, this pest has to come in on cuttings from propagators, making it an excellent pest to be controlled by cutting dips.
As a first step, we tested the efficacy of several biopesticides and biopesticide combinations against whitefly. We found two effective treatments: Dipping infested cuttings in a combination of 1.25 g./L BotaniGard WP and 0.5% insecticidal soap caused 71% mortality of whiteflies, while a dip in 0.1% SuffOil-X killed 69% of whiteflies*.
Rates of insecticidal soap and oil had to be lowered significantly compared to the recommended spray rates to prevent phytotoxicity. Poinsettia is quite a sensitive crop, so other plant species may be able to tolerate higher rates. The dip residues on the cuttings were compatible with whitefly parasitoids Eretmocerus eremicus and Encarsia formosa, so biocontrol can be used after dipping.
In a second series of experiments, we found that the combination of a dip (BotaniGard WP and insecticidal soap) followed by a biocontrol program of either E. eremicus or Amblyseius swirskii provided the best suppression of whiteflies (Figure 2).
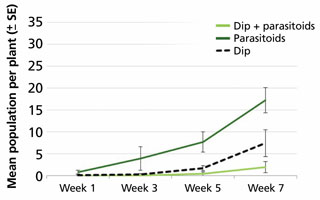 Figure 2. Dips combined with biocontrol resulted in best suppression of Bemisia whiteflies on poinsettia cuttings.
Figure 2. Dips combined with biocontrol resulted in best suppression of Bemisia whiteflies on poinsettia cuttings.
When using only parasitoids (no dips), there was a gradual increase in the number of whiteflies on the plants over 10 weeks. The treatment with only a dip (no parasitoids) had a significant suppressive effect on the starting population and the effects were still present 10 weeks later. Finally, a dip followed by weekly parasitoid releases provided the best results, whereby the dip knocked down the initial whitefly infestation to a level that was kept in check thereafter by E. eremicus. Similar results were obtained when A. swirskii was used in place of E. eremicus.
Commercial greenhouse trials were done in 2015 and 2016 at three locations. Whitefly infestations were very low, so there were no real differences in number of whiteflies between dipped and undipped cuttings. However, it allowed us to look at other factors related to dipping under commercial conditions. No negative effects of dips, such as phytotoxicity or delayed growth, were observed (Figure 3). In a 2016 survey of Ontario poinsettia growers, almost half of the growers reported using cutting dips and 63% reported a reduction in the number of whiteflies.
 Figure 3. No negative effects of dipping were observed (undipped cuttings on the right and dipped cuttings on the left).
Figure 3. No negative effects of dipping were observed (undipped cuttings on the right and dipped cuttings on the left).
For a successful adoption of this technique, cutting dips need to be safe from a phytosanitary point of view. We investigated the risk of disease transfer, using the highly contagious pathogen
Pectobacterium carotovora (Erwinia), which causes soft rot. Although we found low levels of this pathogen (and a few others) in dip solutions at commercial operations, no disease symptoms were observed in the crop.
At Vineland, we only saw disease development when we dipped cuttings in extremely high concentrations of Pectobacterium. Based on these results, we conclude that the level of risk to healthy cuttings appears to be low, but it’s a good practice to change the dip suspension periodically to prevent the potential build-up of pathogens to infectious levels.
To sum up
Large numbers of cuttings can be rapidly treated by dipping, using small amounts of control products. The dip is highly effective, which means that fewer additional control measures are needed for the rest of the growing season and the release of natural enemies can be used as required. By reducing or eliminating the need for pesticides, the issues associated with Bemisia whitefly pesticide resistance are mitigated and there are no re-entry restrictions. Additionally, health risks of greenhouse workers and negative environmental impacts from pesticide sprays or via residues are reduced.
The effectiveness of the “clean start” approach in poinsettia may be transferred to other propagative crops, such as chrysanthemums, mini roses, ivy, etc. While it’s unlikely to be a one-size-fits-all method, it can be a useful tool for early management of other important pests found on cuttings—such as Western flower thrips and two-spotted spider mites—and it also can enhance subsequent biocontrol practices.
Note: Right now, the only product registered for dipping in Canada is Landscape Oil. Label expansions for BotaniGard and Kopa have been submitted. In the U.S., dipping is included as an application method on the label of BotaniGard and label expansion will be submitted soon for Suffoil-X.
GT
Acknowledgments: Researchers and technical staff who participated in the research: Dr. Michael Brownbridge, Dr. Sarah Jandricic, Graeme Murphy, Taro Saito, Mark Jandricic, Ashley Summerfield, Angela Brommit, Paul Côté, Dr. Michelangelo LaSpina, Anna Krzywdzinski, Rebecca Eerkes and Erfan Vafaie.
This project was funded in part through Growing Forward 2 (GF2), a federal-provincial-territorial initiative. The Agricultural Adaptation Council assists in the delivery of GF2 in Ontario. Additional support was provided by Dümmen Orange, BioWorks, Koppert Canada and Flowers Canada (Ontario) Inc. (Guelph, ON, Canada).
*For full details of the study, see Buitenhuis et al. 2016, “How to start with a clean crop: Biopesticide dips reduce populations of Bemisia tabaci (Hemiptera: Aleyrodidae) on greenhouse poinsettia propagative cuttings.” Insects 7 (48).
Rose Buitenhuis is a researcher at the Vineland Research & Innovation Centre in Ontario, Canada.