6/27/2014
Plug & Cutting Conference: Advanced pH Management
Paul Fisher, Ryan Dickson, Bill Argo & Bruce Mackay

The selection of a fertilizer formulation affects whether substrate-pH is likely to drift up or down after planting. Substrate-pH refers to the acidity or basicity of the container growing medium, with a target for most crops between pH 5.6 to 6.4. A low substrate-pH tends to result in iron/manganese toxicity in sensitive crops (Figure 1A), whereas a high substrate-pH results in iron deficiency (Figure 1B). Water alkalinity, which you can think of as dissolved lime in the irrigation water, is one factor that causes substrate-pH to increase over time.
One goal of any fertilizer program is to balance water alkalinity, crop needs and fertilizer selection so that substrate-pH is stable and in the acceptable range over time. In this article, we describe a new calculator tool on the
BackPocketGrower.com website that helps in that decision.
To neutralize water alkalinity, growers may choose to inject acid (such as sulfuric acid). Alternatively, water alkalinity can be balanced with the acidity of the water-soluble fertilizer—high alkalinity is matched with nitrogen (N) in the form of ammonium (acidic effect) or low alkalinity with nitrogen provided as nitrate (basic effect). There are several units of alkalinity: 1 milliequivalent per liter (meq/L) = 50 ppm CaCO3 = 61 ppm HCO3-. An alkalinity between 1.5 and 2.5 meq/L (75 to 125 ppm CaCO3) is in a moderate range.
On many water-soluble fertilizer labels, it will indicate the potential pH effect in “Calcium Carbonate Equivalents.” This might be acidic (meaning the fertilizer will push substrate-pH down over time) or basic (will push pH up). After several years of research at the University of Florida, we have fine-tuned this calculation of the pH effect for water-soluble fertilizers used in soilless container media to improve fertilizer selection.
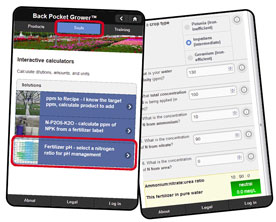
Figure 2 shows how to access the “Fertilizer pH” tool, by going to
BackPocketGrower.com, which will run on smartphones, tablets and desktop computers. Select the “Tools” option at the top of the screen, and choose the Fertilizer pH tool.
Information needed by the Fertilizer pH tool:
- Select the plant species. Petunia is an example of an iron-inefficient crop that tends to increase substrate-pH. Seed or zonal geranium tends to decrease substrate-pH, whereas bedding plant impatiens is intermediate. Ryan Dickson’s Ph.D. research at the University of Florida is screening the pH effect of other species and understanding the physiology of how plant species react with fertilizer.
- Enter the alkalinity of the irrigation water in ppm CaCO3.
- Enter the concentration of N in the fertilizer in parts per million (ppm, which is the same as milligrams per liter). The higher the ppm, the stronger the pH effect of the fertilizer. Nitrogen has the most dominant effect on substrate-pH because more N is taken up than other fertilizer ions, such as phosphorus or potassium.
- Enter the ratio between the three most common nitrogen forms. Ammonium-N is strongly acidic, Nitrate-N is weakly basic and Urea-N is moderately acidic. The balance between these three N forms determines whether the overall pH effect is likely to be acidic, neutral or basic.
- The Fertilizer pH tool calculates the net effect of water alkalinity, plant species and concentration of the different N forms.
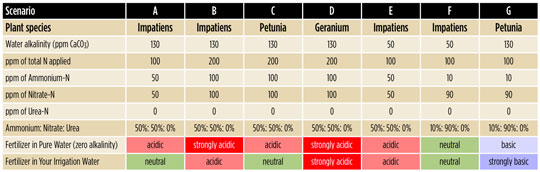
Table 1 shows the effect of the fertilizer in pure water (with no alkalinity) and then combined with the irrigation water alkalinity, with different scenarios:
A. In an impatiens crop, water alkalinity of 130 ppm CaCO3 (2.6 meq/L) is balanced by fertilizer at 100 ppm N containing 50% ammoniacal nitrogen and 50% nitrate
nitrogen.
B. When the fertilizer concentration from scenario A is doubled from 100 to 200 ppm N, the strength of the fertilizer reaction increases, so the overall reaction of the fertilizer solution becomes acidic, causing the media pH to decrease over time. To make the overall reaction neutral at 200 ppm N, you would either have to increase the amount of alkalinity in the water (injecting less acid if acidifying) or use a fertilizer with a lower percentage of ammoniacal nitrogen.
C. With petunias, the same water and fertilizer combination as in scenario B will give a neutral pH reaction because petunia growth is more basic than impatiens growth.
D. With geraniums, the same water and fertilizer combination as scenario B will give a very acidic pH reaction because geranium growth is more acidic than impatiens and much more acidic than petunias. Less ammonium-N would be desirable to avoid a drop in substrate-pH.
E. If the water alkalinity is low (compare with scenario A), then a high ammonium fertilizer will be too acidic and substrate-pH will drop over time.
F. To balance the pH reaction with low water alkalinity, increase nitrate-N and use less ammonium-N or urea-N compared with scenario E.
G. If too many factors are basic—such as this combination of petunia, alkaline water and high nitrate—then substrate-pH will increase over time.
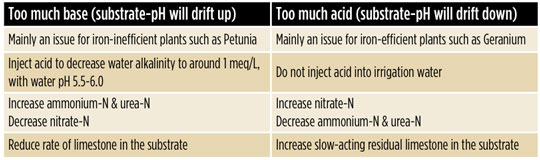
The key is to balance the plant species, concentration of each N form and water alkalinity. Some corrective options are listed in Table 2.
 Conclusion
Conclusion
We encourage you to visit
BackPocketGrower.com and try out the “Fertilizer pH” tool, which was funded by our industry supporters in the Floriculture Research Alliance (
FloricultureAlliance.org) and the USDA-ARS Floriculture and Nursery Research Initiative.
There’s no guarantee that substrate-pH won’t drift up and down even when selecting the correct fertilizer because there are other important factors, such as residual limestone and substrate components. Therefore, regular onsite monitoring of pH and EC is advised to prevent nutritional issues.
GT
Paul Fisher, Ph.D. is Professor and Extension Specialist and Ryan Dickson is a Ph.D. student at the University of Florida IFAS, pfisher@ufl.edu. Bill Argo, Ph.D. is Technical Manager for Blackmore Company, bargo@blackmoreco.com. Bruce MacKay, Ph.D. is Managing Director of thomasBaine Ltd., bruce@thomasbaine.com.