3/22/2011
Fighting Fungus Gnats With Parasitic Nematodes
Julie Graesch
Conventional insecticides have been used to control pests since the early 1900s. In the 1960s, additional options were developed with the establishment of integrated pest management (IPM). Biological control agents (BCAs) are one of the oldest and most effective means for achieving insect control. Although BCAs didn’t catch on right away, leaps and bounds have been made in their acceptances into pest management programs in the past two decades and are now commonly used tools in the pest management toolbox.
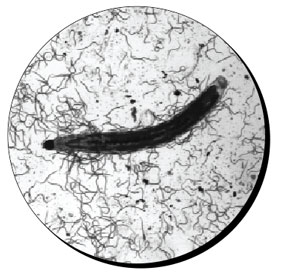
Fungus gnat larvae surrounded by parasitic nematode S. feltiae.
Fungus gnat biology
Fungus gnats are a common and widespread greenhouse pest with cultural, physical, biological and chemical control options that readily lend themselves to IPM programs. Adult fungus gnats are small (1/8 in. long), black-gray flies. Easily monitored with yellow sticky cards, fungus gnat adults have long legs, many-segmented antennae and a single pair of grayish transparent wings with a Y-shaped vein near the wing tip. Adults are weak fliers and often are found near light sources or along the soil surface. Females can lay up to 200 eggs in areas where adults are active. The eggs hatch within three to six days, depending on the temperature, and will then go through four larval instars.
The fungus gnat larvae are the most destructive life stage. Larvae are about ¼ in. long and are translucent, legless and worm-like, with a distinctive shiny black head. Larvae are usually concentrated in the top 3 in. of the soil, but also can be found throughout the soil profile. Fungus gnat larvae prefer to eat fungus, which is needed in their diet for optimal development; however, if fungus is not readily available, larvae will feed on plant material.
Damage and detection
Larval feeding can cause direct root damage, as well as provide an entry into the roots for fungal pathogens like Thielaviopsis, Pythium, Botrytis, Verticillium and Fusarium. Fungus gnat larvae tunnel into thick, succulent stems and roots, causing the plant to wilt and, in severe infestations, die. Seedlings, young plants and unrooted cuttings lacking fully developed root systems are particularly vulnerable to larvae. Larval populations can be monitored by using potato slices, cut 1 in. thick, that are then placed on the surface of growing media. Potato slices should be examined weekly for incidence of fungus gnat larvae.
Fungus gnats thrive in moist, saturated conditions, so cultural practices should be utilized to limit freestanding water and over-saturated growing media. Irrigate only as needed and allow the surface of growing media to dry slightly between waterings. Always repair irrigation system leaks and avoid over-fertilization. Because fungus gnats are weak fliers, physical barriers like mesh screens can be used in vents and doorways to minimize pest movement.
How nematodes can help
If populations of fungus gnats are observed on yellow sticky cards or potato slices that exceed grower-defined action thresholds, biological or chemical control options are warranted. Several BCAs, such as insect parasitic nematodes, predatory mites and beetles, parasitic wasps and beneficial fungi and bacteria can be used to reduce pest populations.
Steinernema feltiae, an insect parasitic nematode, has been used extensively for fungus gnat control for many years.
S. feltiae is a microscopic, non-segmented roundworm that seeks out and enters fungus gnat larvae through natural body openings like the mouth or anus.
S. feltiae uses carbon dioxide, movement and vibrations to locate larvae. Once inside, the nematodes release symbiotic bacteria that quickly kill targeted insects. Reproduction inside the insect releases new generations of infective juvenile nematodes that disperse in search of further prey.
S. feltiae can be used as a stand-alone control option, in combination with other BCAs such as
Bacillus thuringiensis subspecies
israelensis,
Hypoaspis aculeifer, H. miles and
Atheta coriaria, or in rotation with conventional insecticides. Rotation with conventional insecticides affords the opportunity to greatly reduce the number of chemical applications needed for acceptable levels of pest control, reducing the likelihood that pests will develop pesticide resistance.
If using
S. feltiae as a component of a resistance management program, treatments of
S. feltiae should begin early in the growth cycle when pest pressure is low. If new plant material is introduced into a greenhouse area, or if yellow sticky card monitoring indicates that fungus gnat populations exceed action thresholds, conventional insecticides can be used to quickly reduce adult populations. By combining chemicals and biologicals, more stages of the fungus gnats’ life cycle will be targeted, providing more complete pest control.
One of the most effective and easiest ways to apply nematodes is through irrigation injection. Solutions emitting from dosing units can be applied through boom sprayers, overhead and drip irrigation, hand watering and other commonly used watering methods. Nematodes also can be applied through spray tanks, backpack sprayers or by dip applications. When nematodes are in solution, it’s important to ensure stock solutions are constantly agitated to avoid settling in tanks and to prevent nematode solutions from getting above 86F (30C). Air circulation, mechanical circulation and hand-mixing can be used to prevent settling.
Applications of nematodes should be made to moist soil and applied in the early morning or late evening to avoid high temperatures, UV exposure and nematode desiccation. Remove sprayer filters of 50 mesh or finer and prevent pump pressures from exceeding 300 psi. If nematodes are applied over foliage, irrigate immediately with plain water to wash nematodes off the foliage and into the soil. Soil temperatures should be between 50F and 86F (10C and 30C) in order for nematodes to actively seek insect hosts.
To learn more about nematode circulation, chemical compatibility, application rates through irrigation injection, and other important application information, please visit
www.beckerunderwood.com, call (515) 232-5907 or visit our new beneficial nematodes blog at
www.beckerunderwood.com/blog.
GT
Julie Graesch is a nematode field development specialist at Becker Underwood, in Ames, Iowa, where she is responsible for product development, as well as coordinating research opportunities with universities and third parties. Julie can be reached at julie.graesch@beckerunderwood.com.